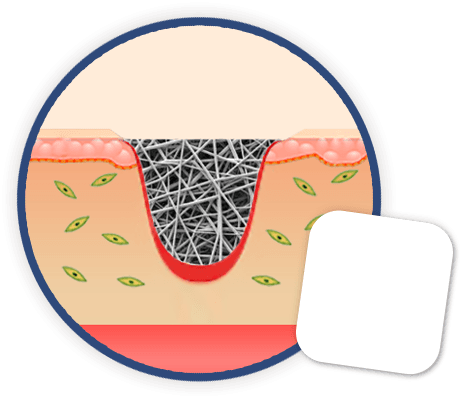
EktoTherix™
EktoTherix™ is made up of electrospun fibres at the correct scale for cell attachment and movement into the wound space.
Benefits:
- Bioresorbable
- Degrades by hydrolysis and is resorbed during the healing process
- Less intervention and disturbance (as repeated dressing changes will not be required)
- Aims to improve quality of the repair (less scar, granulation tissue formation promoted, better correction of contours, better overall cosmesis)
Achievements
Completed First in Man (FIM) Clinical Investigation for surgical excisional wounds at 3 hospital sites and our 15 patient target achieved
The FIM trial established EktoTherix™ is safe to use for the treatment of acute wounds - created by the surgical removal of non-melanoma skin cancers in this case.
For some patients healing time was decreased and a better cosmetic outcome was achieved.
Promotion of granulation tissue formation observed in FIM – suggesting additional applications for EktoTherix™
Case study images from EktoTherix™ First in Man Clinical Investigation

Pre-surgical (squamous cell carcinoma, SCC)

After excision of SCC but prior to EktoTherix™ application

After EktoTherix™ implantation

Three months after surgery
Get in touch with us
We’re always happy to hear from potential investors and funding partners as well as researchers, universities and networks interested in our products and development work.
